Medtronic announced CE mark approval and the first commercial European implants for its OmniaSecure system.
The OmniaSecure defibrillation lead connects to an implantable defibrillator. It can treat potentially life-threatening ventricular tachyarrhythmias, ventricular fibrillation (VT/VF) and bradyarrhythmias. Built on the company’s SelectSecure Model 3830 pacing lead for catheter delivery, it adds to the wide-ranging Medtronic portfolio of lead solutions for precise delivery and placement.
Related: Anumana secures FDA clearance for ECG-AI algorithm
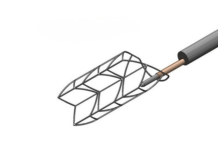
Medtronic labels it the world’s smallest defibrillation lead, coming in at 4.7 Fr, or 1.6 mm. That equals about the diameter of graphite in a wooden pencil. The defibrillator lead won FDA approval for right ventricle placement in April 2025 and launched at the start of this year. It then picked up expanded FDA approval for conduction pacing last week.
The medtech giant said that OmniaSecure earned European approval for traditional placement within the right ventricle.
According to Medtronic, leads sense the heartbeat and transmit signals to the implanted pacemaker device, which then delivers therapy. Transvenous leads remain vulnerable due to potential degradation and fracture. Current options often come in larger in diameter than OmniaSecure, which Medtronic said can lead to complications like venous occlusion or tricuspid valve regurgitation.