Inceptiv is claimed to be the only SCS system in Europe that provides both full-body 1.5T and 3T MRI access.
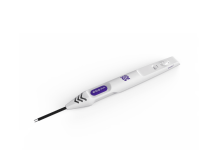
Medtronic has secured CE Mark approval for its closed-loop rechargeable spinal cord stimulator (SCS), Inceptiv, to treat chronic pain.
Inceptiv is claimed to be the company’s first SCS device to provide a closed-loop feature for sensing the unique biological signals of the individual.
It also helps in adjusting stimulation moment to moment as per the requirement, ensuring that therapy aligns smoothly with the motions of daily life.
The device deploys a closed-loop feature using Evoked Compound Action Potentials (ECAPs) to perceive and react to signals along the spinal cord.
Inceptiv can sense the body’s neural response to stimulation 50 times every second, all day.
It is also claimed to be the only SCS system in Europe that provides both full-body 1.5T and 3T MRI access.
According to the company, at just 6mm, Inceptiv is the thinnest SCS device in the world and provides the benefits of DTM spinal cord stimulation.
Medtronic Neuromodulation unit chief medical officer Ash Sharan said: “Medtronic pioneered SCS for pain management more than 50 years ago, and we continue to deliver new innovations that personalise care and improve pain relief for patients.
“This approval marks the beginning of a new era of pain relief, using sensing technology to listen to the unique biological signals of each patient.”
The Inceptiv SCS system is set to become available for purchase in Europe in the next few months. It has not yet received approval for sale or distribution in the US.