The EyeBOX is the First FDA-cleared Objective Test for Concussion
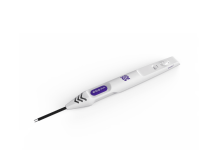
Oculogica, a leader in concussion diagnostics, announced launch of the new EyeBOX device following FDA clearance earlier this year. The latest EyeBOX is lightweight, battery-operated, and small enough to fit in a backpack, making it easy to use in both clinical and sports settings.
The EyeBOX is the first objective diagnostic cleared by the FDA as an aid in the diagnosis of concussion. Using a proprietary algorithm, the EyeBOX test takes less than 4 minutes. The test is non-invasive, does not rely on the patient’s literacy level, and can be used in both pediatrics and adults, for injuries due to any type of activity.

The latest EyeBOX is lightweight, battery-operated, and small enough to fit in a backpack.
Concussion is a form of mild traumatic brain injury (mTBI), caused by an external force to the head or body resulting in disruption of normal brain function. According to the Centers for Disease Control (CDC), concussions accounted for approximately 2.5 million emergency visits in 2014 in the United States.
“With increasing rates of concussion, particularly in adolescents, the need for an objective diagnostic that isn’t dependent on patient-reported symptoms is critical to ensuring proper treatment and management to prevent further injury,” said Christina Master, MD, FAAP, CAQSM, FACSM, pediatrician and sports medicine specialist in brain injury at Children’s Hospital of Philadelphia (CHOP) . “Earlier identification of these abnormalities allows clinicians to improve diagnosis capabilities and individualize recommendations for activity as well as return to work or school.”
In collaboration with clinicians and researchers at leading US institutions, the EyeBOX device has been clinically validated in multi-center studies and has been included in numerous peer-reviewed medical journals.
“Our EyeBOX device offers a leap forward in concussion diagnosis. We are particularly excited to be launching this during Brain Injury Awareness Month,” said Rosina Samadani, PhD, CEO of Oculogica. “The lightweight form factor and battery operation allow it to be easily transported. Concussions happen everywhere and our latest device was designed with that in mind.”
About Oculogica
Oculogica, Inc. is an eye tracking-based neuro-diagnostic company founded by Dr. Uzma Samadani, Associate Professor of Neurosurgery at the University of Minnesota. The company has developed the EyeBOX, the first baseline-free, aid in diagnosis of concussion to help physicians objectively evaluate patient. The test is less than 4-minutes and appropriate for ages 5 to 67 years. To learn more, visit www.oculogica.com.