Starlight Cardiovascular announced that the first baby in the world received treatment with its Lifeline pediatric stent system.
The first implant, performed by Dr. Shabana Shahanavaz, took place at Cincinnati Children’s Hospital in January. It occurred as part of an FDA investigational device exemption (IDE) clinical trial. Shahanavaz performed the procedure on an eight-day-old baby with double inlet left ventricle and pulmonary atresia, a complex congenital heart defect that results in inadequate blood flow to a baby’s lungs.
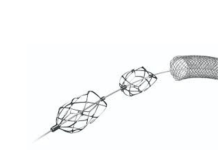
San Diego-based Starlight designed its Lifeline stent to keep the ductus arteriosus open. This vessel exists in the fetal circulation and closes shortly after birth. Keeping it open provides critical pulmonary blood flow in newborns.
According to Starlight, congenital heart disease is the most common type of birth defect, affecting 1% of all newborns. Babies with certain CHDs require immediate intervention to survive, in some cases needing open-chest surgery. Lifeline provides a minimally invasive alternative to this major surgery for newborns, the company says.
Shahanavaz said:
“In my 15 years of practice, we have adapted adult-designed stents to treat newborns because no dedicated alternative existed. The Lifeline stent is a major step forward — its design, flexibility, and precision make it substantially better suited for neonatal ductal anatomy. This first-in-human implantation highlights how purpose-built technology can transform care for our smallest patients. We are especially excited and proud to be the first site to implant this novel stent, underscoring our commitment to advancing innovation and bringing the latest, most thoughtfully designed technologies to the patients who need them most.”
Dr. Aimee Armstrong, national principal investigator and director of the Cardiac Catheterization Laboratory at Nationwide Children’s Hospital, said:
“Up until the availability of the Lifeline stent in this trial, babies needing an open ductus for blood flow to the lungs have either been getting a high-risk surgery or a ductal stent approved only for adult heart disease. These adult stents are stiff and not made for highly curvy ducts. The Lifeline stent is purpose-built for ducts, which turn and bend, sometimes in a 360-degree configuration. We are so happy to start using the Lifeline stent in this trial, as it has the potential to be much safer and easier to use than the current adult stents.”