The SI joint implant uses Nanotex surface technology to improve bone growth.
SurGenTec has secured approval from the US Food and Drug Administration (FDA) for its TiLink-L sacroiliac (SI) joint fusion system.
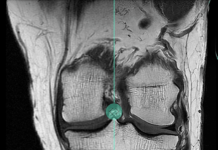
The SI joint implant can be implanted using a lateral or posterior/oblique approach. It leverages the company’s Nanotex surface technology to enhance bone growth.
TiLink-L is the company’s first implant in the sacroiliac products portfolio.
It features an intuitive design that facilitates better compression across the joint, serving as an advanced platform for fusion.
SurGenTec CEO Travis Greenhalgh said: “We are thrilled to announce the FDA clearance of our first treatment option in our sacroiliac family of products.
“With its unique properties and ability to adapt to diverse patient needs, TiLink-L is set to offer physicians the ability to stabilise the sacroiliac joint in a variety of approaches.
“Whether in an outpatient or hospital setting, we believe this device is poised to make a significant difference in patient care.”
TiLink-L also has a helical self-harvesting channel that captures the patient’s own bone and promotes healing.
It also enables graft windows to be strategically positioned between threads to facilitate potential fusion through the screw to improve stability.
The company is offering the implant in several lengths to ensure a custom fit for each patient.
In-vivo studies with the system showed that it promotes bony in-growth and on-growth, which can improve stability for enhanced surgical outcomes.