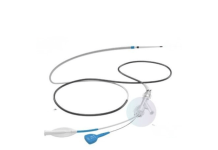
The neurostimulation device, which is currently for investigational use only, aims to treat cognitive and functional decline in Alzheimer’s patients.
Cambridge-based Sinaptica Therapeutics, a company providing electromagnetic treatment for patients with neurodegenerative diseases, announced its receipt of FDA Breakthrough Device Designation for its non-invasive SinaptiStim – AD System, designed to treat cognitive and functional decline in Alzheimer’s patients.
SinaptiStim uses neurostimulation and brain wave monitoring technologies with an AI-personalization engine to provide electromagnetic therapy for cognitive and functional decline in Alzheimer’s patients.
The FDA designation is based on outcomes from a Phase II clinical trial involving mild-to-moderate Alzheimer’s patients that has yet to be published in a peer-reviewed journal.
The System is for investigational use only. The FDA designation expedites the device’s development, assessment and review. It also gives Sinaptica priority review and interactive communication through the premarket review process with the FDA.
“This marks an important milestone for the company, as it helps establish our regulatory pathway for FDA clearance of our SinaptiStim – AD System,” Rich Macary, president of Sinaptica Therapeutics, said in a statement. “We plan to continue working toward the initiation of our pivotal trial next year, as well as further advancing the emerging field of electromagnetic therapeutics given their unique ability to modulate key mechanisms of synaptic plasticity and network connectivity in targeted regions of the brain.”
RELATED: Medtronic’s Hugo surgical robot collects green lights in Europe, Canada, Japan
THE LARGER TREND
Other companies are utilizing neurostimulation, the purposeful modulation of specific portions of a patient’s brain, spinal cord or peripheral nervous system, to treat various other conditions.
DyAnsyus, a Calif.-based medical device company specializing in the autonomous nervous system, offers Primary Relief, a percutaneous electrical nerve stimulator (PENS) system to treat postoperative pain following cardiac surgery. Last month, the PEN system received FDA approval.
Publicly-traded NeuroPace develops implantable devices to treat neurological disorders. Its RNS system is a neurostimulation therapy device for adults who experience frequent and disabling epileptic seizures.
In August last year, NeuroPace launched its nSight Platform. The portal gathers information about a patient’s seizures and health progress to provide doctors data points to personalize care, not long after the company went public.
Are you Hiring?