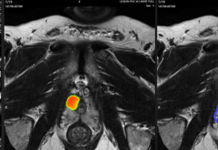
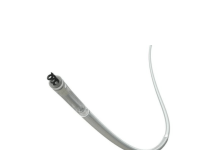
Serenity Medical announced that it received FDA Humanitarian Device Exemption (HDE) approval for its novel River stent.
Harrison, New York-based Serenity said approval makes River the first FDA-approved cerebral venous stent. The company specifically developed its technology for the treatment of severe idiopathic intracranial hypertension (IIH) in adult patients who have failed medical therapy.
Related: Medtronic wins CE mark for OmniaSecure pacemaker lead
IIH, caused by elevated intracranial pressure, leads to debilitating chronic headaches, vision loss and cognitive impairments. The company says it remains difficult to find appropriate, FDA-approved surgical therapies to safely relieve its symptoms.
According to a news release, the approval marks a landmark clinical milestone for the purpose-built tool. It helps address a condition that has “long challenged the neurovascular community and suffering patients,” Serenity said.
The company supported its approval with findings published in the Journal of Neurointerventional Surgery (JNIS) in February 2025. It enrolled 39 subjects across five U.S. centers, meeting its primary safety endpoint with an observed major adverse event rate of 5.4%. Investigators saw improvements in opening CSF pressure, headaches, papilledema, pulsatile tinnitus, visual symptoms and quality of life scores.
Serenity Medical said it formed a strategic partnership with Radical Catheter Technologies to commercialize the River stent. Humanitarian Device Exemption enables the use of River in adults with IIH with significant stenosis who are resistant or intolerant to medications.
Dr. Y. Pierre Gobin, founder of Serenity Medical, said:
“Delivering the first venous stent approved for severe, refractory IIH is incredibly meaningful to our dedicated team and to people living with IIH who have had limited options for relief until now. Our company was founded with the goal of addressing a problem that has long perplexed the medical community and caused debilitating symptoms in the women suffering with it. Reaching this moment reflects years of persistence, partnership with investigators, and a shared commitment to bring new possibilities to this underserved patient community.”