Endovascular Engineering (E2) announced that it raised $80 million in an oversubscribed Series C financing round.
Gilde Healthcare and Norwest co-led the latest financing for the Menlo Park, California-based company. Existing investors, including Santé Ventures, 415 Capital, S3 Ventures, Panakès Partners, M&L Healthcare Investments, also participated. Two existing undisclosed strategic investors and a new global strategic investor took part, too.
Related: Route 92 Medical brings in $50M growth financing
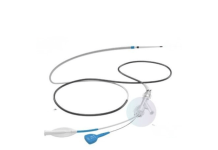
E2 said the latest funding supports commercialization efforts for its novel Hēlo thrombectomy platform. The company developed Hēlo to treat pulmonary embolism (PE).
According to E2, it designed Hēlo to address the limitations of legacy systems for treating PE. Its technology features a patented dual-action mechanism that integrates aspiration with mechanical clot disruption. Using a small-profile catheter, the system delivers large-bore performance to enable efficient single-pass procedures.
E2 says it developed its system to support procedural efficiency and clinical performance as the adoption of mechanical thrombectomy increases. Current mechanical thrombectomy leaders include Stryker after its acquisition of Inari Medical, as well as Boston Scientific ahead of its planned acquisition of Penumbra. Other thrombectomy technology players include the likes of Imperative Care, Inquis Medical and Jupiter Endovascular.
“This is a pivotal milestone for E2 as we continue our mission to improve options for patients and physicians in the VTE space,” said Dan Rose, chief executive officer at E2. “We are incredibly proud of the capabilities of the Hēlo® Platform, and this financing enables us to build a strong commercial organization while continuing important investments in R&D and Operations.”