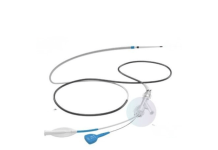
CorTec GmbH, a pioneer in fully implantable brain-computer interfaces (BCI), announced that the U.S. Food and Drug Administration (FDA) has granted Breakthrough Device Designation for the company’s Brain Interchange system. The designation covers the use of direct cortical electrical stimulation to support motor recovery in people living with chronic stroke-related impairments.
The Breakthrough Device Designation is granted to medical technologies that have the potential to provide more effective treatment for life-threatening or irreversibly debilitating conditions. The program is designed to facilitate medical device development and review through prioritized and more interactive engagement with the FDA.
Related: FDA grants 510(k) clearance for Allevion Medical’s Vantage system
CorTec’s Brain Interchange system combines neural signal recording with adaptive stimulation in a closed-loop system. The platform is currently being evaluated in an FDA-approved IDE study at the University of Washington in Seattle, marking the first clinical investigation of a fully implantable, wireless BCI system for stroke rehabilitation in humans.
Stroke is the leading cause of acquired long-term disability in adults worldwide. Approximately 9 million ischemic strokes occur globally each year, with an estimated 1.7 million cases in the United States and Europe alone. Over 80% of stroke patients experience upper-limb impairment, and approximately 50% remain permanently disabled despite standard rehabilitation. For chronic stroke patients with moderate-to-severe motor deficits whose recovery has plateaued after conventional therapy, no approved implantable treatment exists today.
“Only a few BCI companies worldwide – including Neuralink, Synchron, or Blackrock Neurotech – have received Breakthrough Device Designation to date. Achieving this designation is a defining milestone for CorTec and underscores the potential of our Brain Interchange system to address the significant unmet need in stroke rehabilitation,” said Dr. Frank Desiere, CEO of CorTec. “Together with promising initial results from our first-in-human study in Seattle as well as additional long-term data published in Nature Scientific Data1 demonstrating signal stability over 500 days, this designation provides strong momentum as we advance toward larger clinical trials. We believe CorTec occupies a unique position in the global BCI landscape, combining a fully implantable, bidirectional closed-loop platform with a therapeutic approach focused on restoring motor function after stroke.”
CorTec’s approach represents a distinct direction within the global BCI field. While a number of BCI systems have received Breakthrough Device Designation and are restricted to enable patient communication by controlling digital devices through thought, the Brain Interchange™ platform is being developed as a fully implantable, bidirectional device, enabling brain-based communication but also therapeutic neurostimulation, e.g. allowing to restore motor function after stroke. At present, no other BCI company worldwide holds a Breakthrough Device Designation for this indication.
“The Breakthrough Device Designation enables more frequent and structured engagement with the FDA as we advance our development program,” said Mara Assis, Head of Regulatory Affairs & Quality Management at CorTec. “Our regulatory strategy has followed a stepwise approach, from prior device clearance to IDE approval and successful human implantations. This designation will help accelerate planning of clinical trials and support the next regulatory milestones.”
CorTec continues to advance the Brain Interchange System as a flexible, adaptive platform with potential applications across multiple neurological conditions. In addition to the ongoing stroke rehabilitation study, the Brain Interchange platform is being evaluated for epilepsy, with further indications including paralysis and depression under development.