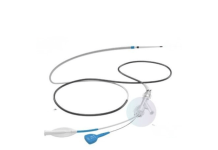
ATRO Medical, the Dutch clinical-stage orthopedic company developing a novel artificial meniscus implant for patients suffering from post-meniscectomy pain, announces the successful closing of a €3 million financing round. The financing round is primarily composed of equity investments, complemented by non-dilutive funding. It brings together MedTech investors from the Netherlands, Switzerland, and the United Arab Emirates such as 819 Capital, Thuja Capital, Bolwork International Investment and industry leaders DSM-Firmenich Ventures and Samaplast.
The funding comes at a decisive moment in the company’s development. ATRO Medical has reached an important milestone in the clinical evaluation of its latest Artimis® Meniscus Prosthesis design. The ongoing studies are designed to further assess the artificial meniscus system in patients experiencing persistent knee pain following meniscus removal.
In addition to its clinical progress, ATRO Medical has strengthened its intellectual property position and gained international traction by its selection for the prestigious PlugAndPlay Health incubator in the US, positioning the company for the next phase of growth.
The newly secured capital will be used to expand clinical evidence generation in ongoing studies, prepare the company for the pivotal clinical trial in both the United States and Europe, and further optimize manufacturing, regulatory readiness, and clinical operations.
Michiel Schwartz, Chairman of the Board of ATRO Medical, comments: “This investment reflects strong international confidence in ATRO Medical’s technology, team and strategy. The recent clinical outcomes confirm that we have cracked the code for patients with post-meniscectomy pain. With this experienced leadership team and a clear path toward pivotal studies in the US and Europe, ATRO Medical is entering a defining phase in its development.”
As part of this investment round, ATRO Medical announces a strategic leadership transition:
- Maarten van der Zanden has been appointed Chief Executive Officer, transitioning from his previous role as Chief Implementation Officer. He is a seasoned MedTech marketing executive with a proven track record of leading successful international product launches across multiple medical specialties.
- Anita Brinks continues in her role as Chief Operating Officer, overseeing operations and quality systems as the company prepares for pivotal clinical programs. She brings over 25 years of experience in medical practice and healthcare innovation, including senior executive leadership positions, providing a strong foundation for operational excellence and strategic execution.
- Founder Tony van Tienen will focus on clinical strategy, surgeon engagement and scientific development as Chief Medical Officer. In addition to his responsibilities at ATRO Medical, he practices as knee surgeon at the Viasana Clinic and is internationally recognized for his expertise in meniscus surgery and joint preservation.
This executive structure is designed to align market preparation, operational execution, clinical strategy and fund raising, as the company advances toward regulatory milestones.
Following strong interest from private investors and patients, ATRO Medical will extend the current financing round through a share funding campaign on the Eyevestor platform. Building on the secured €3 million, this initiative enables individuals to participate in the company’s next phase of clinical development and international expansion at attractive company valuation. By opening the round to a broader community, ATRO Medical aims to provide patients and private investors the opportunity to actively contribute to advancing the future of meniscus surgery. The campaign will launch in March on the Eyevestor website.
Hundreds of thousands of patients worldwide experience persistent knee pain after meniscus removal, often combined with progressive osteoarthritis, at a relatively young age without a suitable surgical treatment at hand. ATRO Medical’s artificial meniscus is designed to restore joint biomechanics and relieve pain while preserving future treatment options.
With this financing round, the company is positioned to accelerate clinical development and move closer to offering a durable, joint-preserving solution for patients in need.