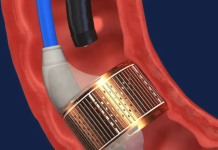
Airiver Medical announced that it received FDA breakthrough device designation for its pulmonary drug-coated balloon (DCB).
Additionally, Airiver announced the first treated patients in a clinical trial for the DCB.
The Brooklyn Park, Minnesota-based company designed its DCB to treat central airway stenosis. Central airway stenosis (airway narrowing) can lead to prolonged intubation, tracheostomy, stenting, tuberculosis or lung transplant.
Related: Qure.ai nets six new indications cleared by the FDA
Airiver developed its DCB to combine standard balloon dilation with proprietary drug delivery. It aims to open the respiratory tract and deliver therapeutics designed to maintain symptom relief and prevent recurrence. The system’s proprietary coating enables very localized paclitaxel delivery to the stenosis while limiting levels in the surrounding healthy tissue.
In August, 2025, the company received FDA investigational device exemption (IDE) for the DCB, enabling the launch of a pivotal trial. It now has a breakthrough nod as it treats the first patients in the study.
“Receiving this designation is extremely meaningful for us because with it, patients and health care providers may gain more timely access to our novel DCB technology, with the potential to provide safer and more effective treatment,” said Mitchell Erickson, director of R&D for Airiver Medical. “The Airiver DCB has the potential to fill a gap that currently exists, as there is no optimal treatment of recurrent airway stenosis available as part of today’s treatment paradigm.”
Dr. Ashli O’Rourke, professor and director of laryngology at the Medical University of South Carolina, treated the first patient. It comes as Airiver looks to enroll up to 200 patients suffering from central airway stenosis. The study compares the Airiver DCB to the standard of care, balloon dilation.
If successful, the company expects the study to serve as the basis for its regulatory submission to the FDA, eventually supporting commercialization in the U.S. The device currently remains investigational.
“Central airway stenosis is a debilitating condition with no minimally invasive, long-lasting treatment,” said Dr. O’Rourke. “This technology has the potential to provide a life-changing treatment option. That’s why I’m thrilled to treat the first patient in this potentially groundbreaking clinical trial.”