The launch of the new analyser comes after it received approval from the US FDA.
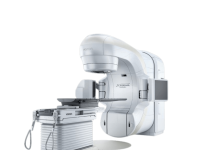
Siemens Healthineers has announced the launch of the new Atellica CI Analyser for clinical chemistry and immunoassay testing.
The launch comes after the analyser received approval from the US Food and Drug Administration (FDA). It is now available in several countries across the world.
The Atellica CI Analyser enables both standalone labs and satellite labs within larger health networks to access the same reagents, consumables and intelligent software found in the company’s Atellica Solution.
All of this has been condensed into a compact 1.9m² footprint, making it suitable for smaller laboratories.
It supports labs to offer more predictable sample turnaround times by enabling random access sampling, micro-volume aspiration and automatic maintenance and quality control scheduling.
RELATED NEWS: CMR Surgical Launches Versius Trainer in VR
Chemistry and immunoassay engines operate independently, ensuring that throughput remains unaffected even if either of the two needs to stop.
Furthermore, the analyser leverages the Atellica Laboratory Evaluation Suite for delivering inspection-ready reports.
Each lab in a health network has a menu of over 200 assays across 20 disease states and can select the best tests for its patients.
More than 50 assays hold the potential to deliver results within 14 minutes.
Siemens Healthineers Diagnostics head Sharon Bracken said: “Labs today need testing instrumentation and informatics that can easily keep pace with rapidly changing testing demands as they occur.
“This next-generation laboratory analyser anticipates workflow bottlenecks, mitigates them proactively, and delivers insights based on data that help laboratory staff do their job more effectively and efficiently.”