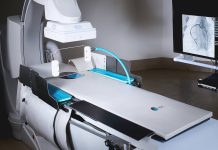
US-based Noah Medical has published positive data from a clinical study demonstrating the success and safety of its Galaxy surgical robotic system.
The results were published in the Asian Pacific Society of Respirology journal and showed that the robotic platform achieved 100% successful navigation and 100% tool-in-lesion accuracy.
Related: NXTSTIM’s wearable nerve stimulation device helps manage long-term pain
The first-in-human FRONTIER study enrolled 18 patients with moderate-risk peripheral pulmonary nodules in Sydney, Australia. The data demonstrates the Galaxy system’s capability to safely access peripheral lung nodules with bronchoscopy – a procedure used to examine and treat the inside of the lungs using a flexible tube with a camera.
Noah Medical’s Galaxy system is used to improve location accuracy and diagnosis of lung nodules. It is incorporated with TiLT+ [Tool in lesion] technology with integrated tomosynthesis and augmented fluoroscopy. It comes with a single-use disposable bronchoscope to enhance procedural workflow and has a small, compact footprint to fit in even small bronchoscopy suites.
The company received US Food and Drug Administration (FDA) clearance for the Galaxy system in March 2023. Shortly after, the medical robotics company secured $150m in a Series B funding round to scale its endoluminal robotic solutions. The oversubscribed financing round was led by Softbank Vision Fund and co-led by Prosperity7 Ventures. The first procedure was conducted in the US in May 2023, marking the platform’s commercial release.
Noah Medical’s CEO Jian Zhang said: “After years of working with leading pulmonologists to understand and address the challenges of diagnosing lung cancer with existing technologies, the Galaxy System’s groundbreaking technology makes it easier than ever to locate and diagnose this deadly disease.”