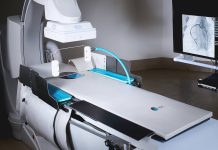
Microbot Medical announced the completion of the first procedure in a clinical trial using its surgical robot system. Investigators in the company’s pivotal human clinical trial completed the procedure at Brigham and Women’s Hospital in Boston. They used the Braintree, Massachusetts-based company’s Liberty endovascular robotic surgical system.
Dr. Dmitry Rabkin, the trial’s lead investigator, performed the first human case at BWH. The trial is part of the company’s FDA investigational device exemption for Liberty, which it announced last month. Microbot Medical expects to use the results to support future FDA submissions and subsequent commercialisation.
Related: Vectorious Medical reports positive data from study of miniature heart sensor
The company designed its Liberty surgical robot for potential use in neurovascular, cardiovascular and peripheral vascular procedures. The system includes a compact design and remote operating capabilities to reduce radiation exposure and physical strain on physicians. Microbot believes Liberty’s remote operation could potentially make it the first system to democratize endovascular interventional procedures.
“Enroling the first patient in our pivotal human clinical trial is a significant milestone for the company, and an important step on our journey to bring Liberty to U.S. physicians,” said Dr. Juan Diaz-Cartelle, chief medical officer of Microbot Medical. “We are very pleased with the rapid pace of site activation, and I’m looking forward to enroling additional patients in the near future.”
Recent highlights for the company include expanded U.S. operations ahead of the IDE submission to prepare for the next steps. Microbot already had momentum after positive results from its good laboratory practices (GLP) pre-clinical study. The company also secured a $2.154 million settlement of a lawsuit involving a securities purchase agreement in January.